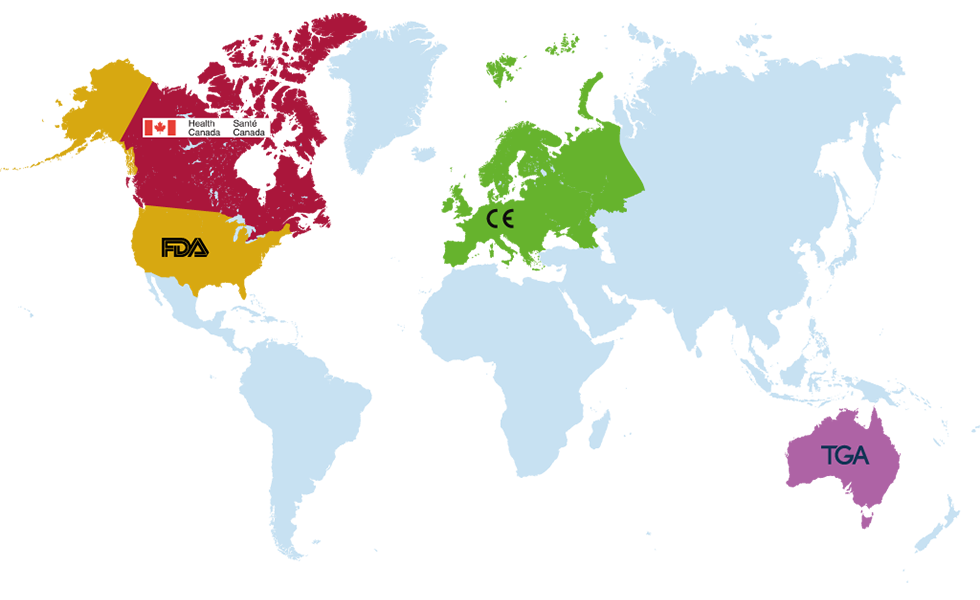
Kinesis Gait™ is a registered medical device in Europe, United States, Canada and Australia.
It is certified for electrical safety as a medical device to EN 60601-1
Clinical grade Gait Analysis, validated through fourteen years of research.
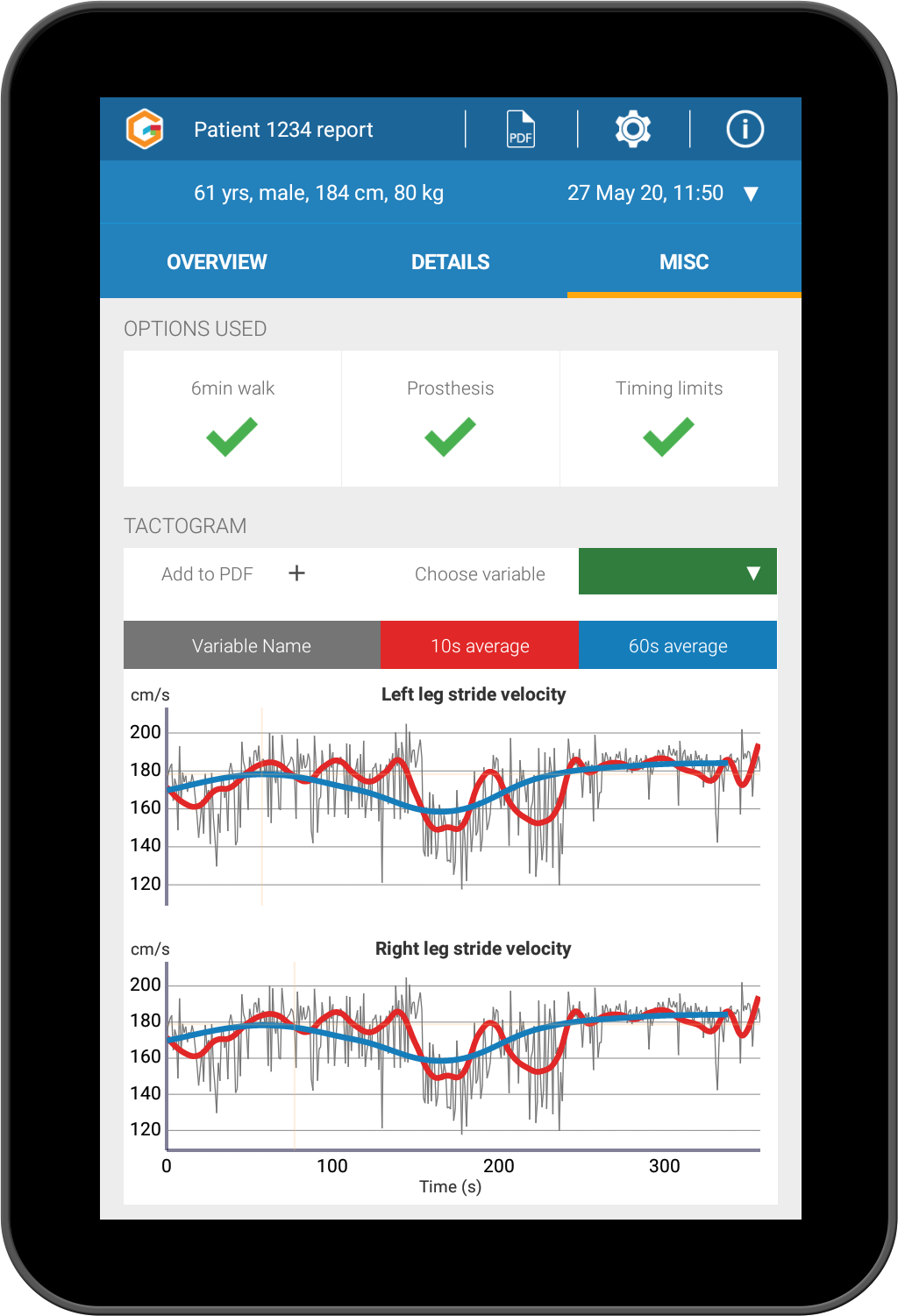
Proven and reliable measurement of gait. 89 different bilateral gait measures: temporal and spatial gait parameters as well as
gait variability, gait symmetry, raw sensor data and individual stride data.
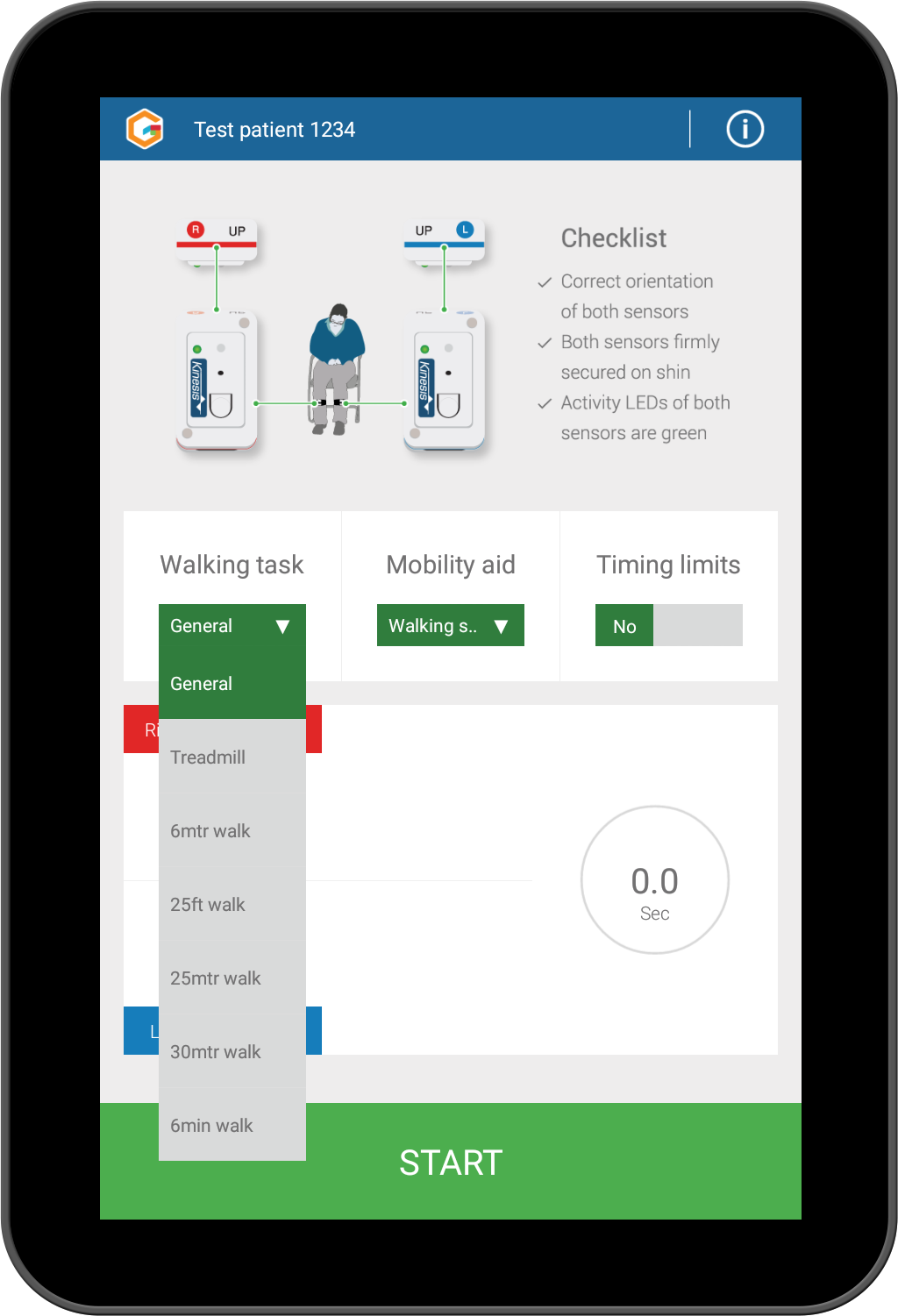
Record data for multiple walking tasks (including dedicated 6 minute walk mode).
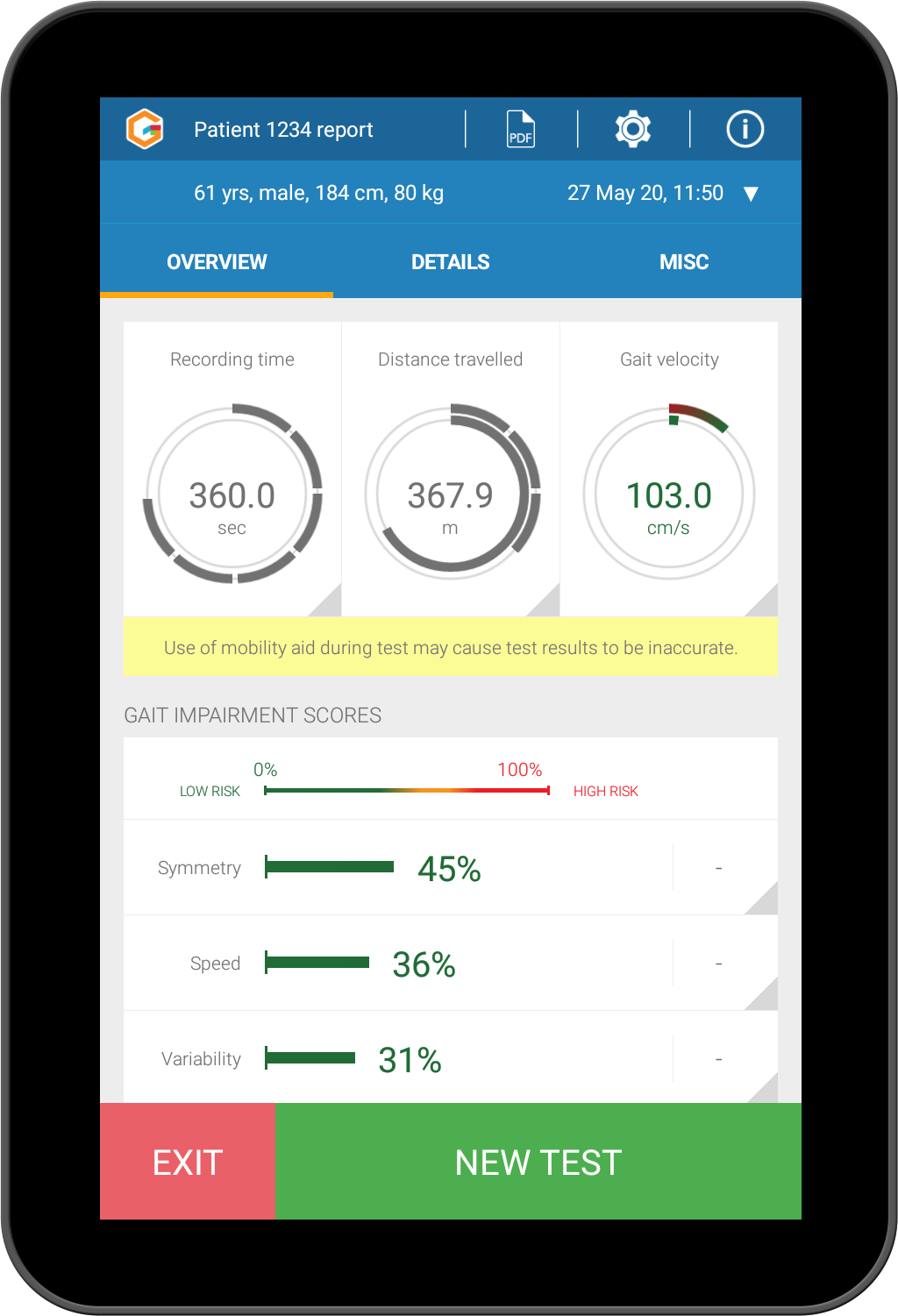
Identify gait impairment and gait change
Kinesis Gait™ uses gait analysis to identify specific gait and mobility problems
using percentile scores (based on speed, gait variability, gait symmetry) calculated from an age and gender matched reference data set of over 1,400 patients.
The results are colour-coded to highlight mobility impairment.
Proven, validated gait analysis technology
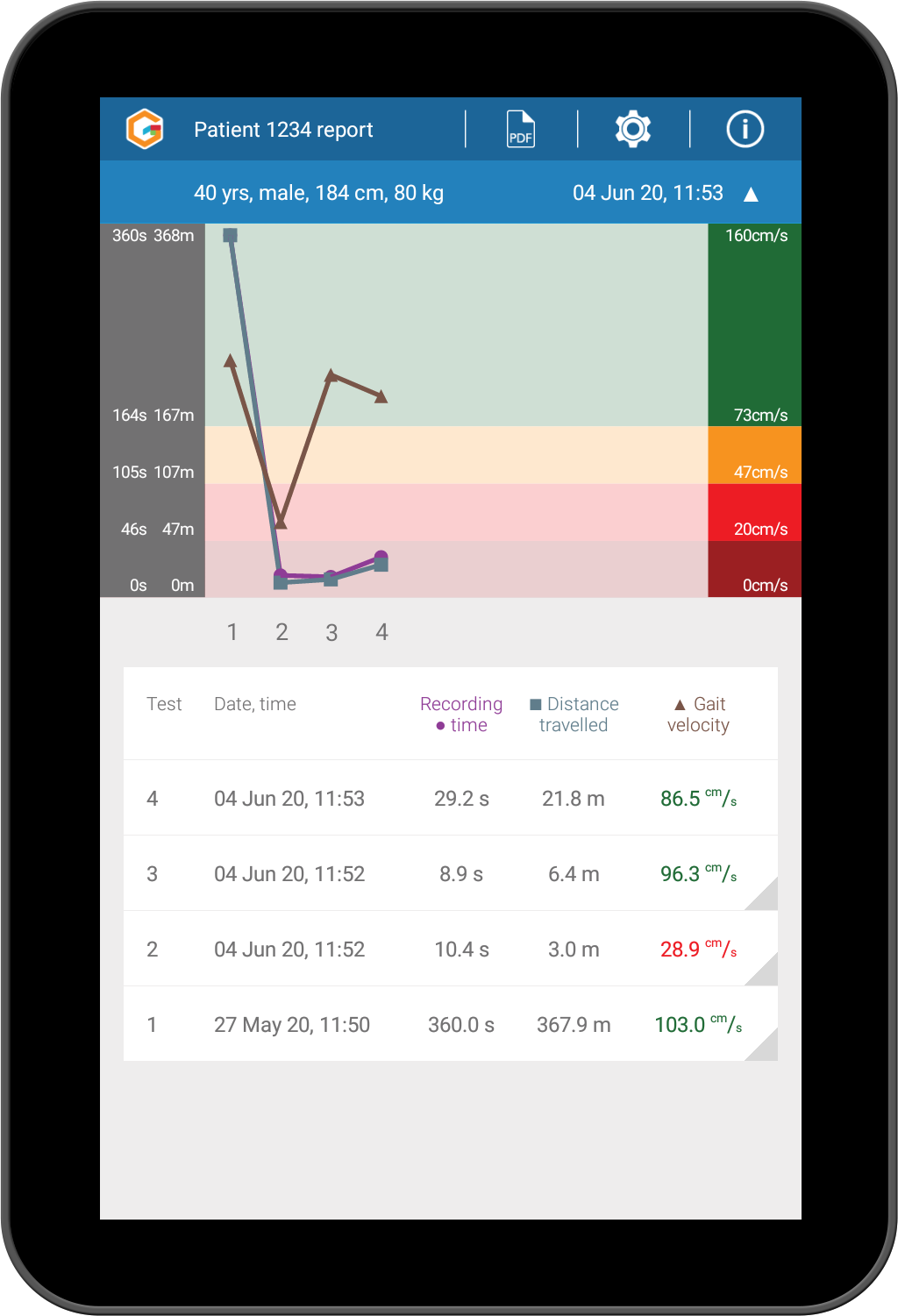
Gait as a digital biomarker of disease. Measure how well a patient responds to intervention, therapy or medication by trending gait data over time, as well as viewing all historical tests. 89 validated parameters for measurement of gait.
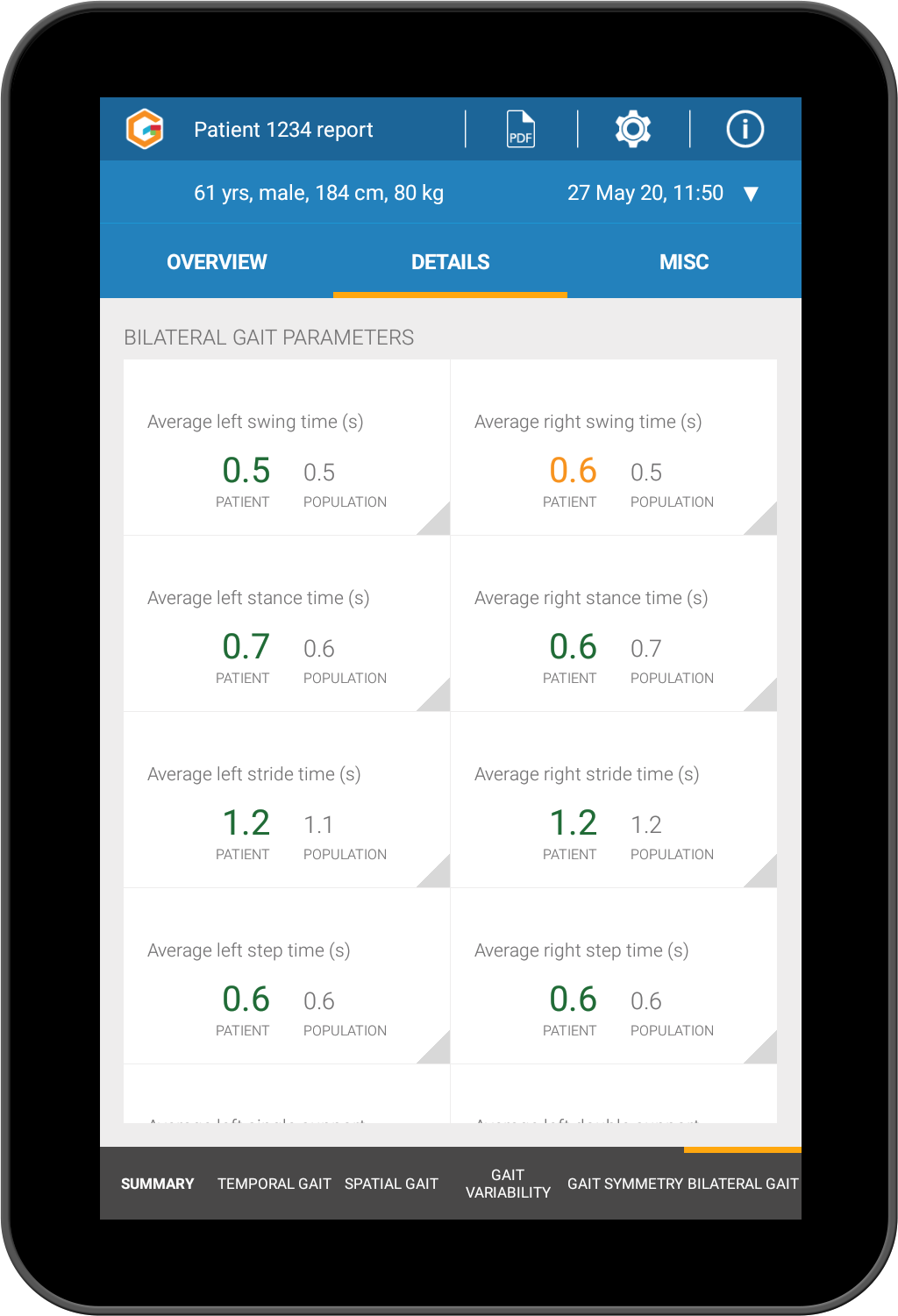
Objective assessment of each element of gait including, individual stride analysis, bilateral gait, spatial gait parameters, temporal gait parameters, gait variability and gait symmetry.
Customise results by pinning most relevant data to the summary results screen.
Compare against normative data - Data are colour-coded to highlight any statistical deviations from reference values.
Intuitive display of results; results are available immediately upon completing a test and can be immediately backed up to the cloud. Ability to track use of mobility aids, walking tasks and test conditions.
Prompt and friendly customer support.
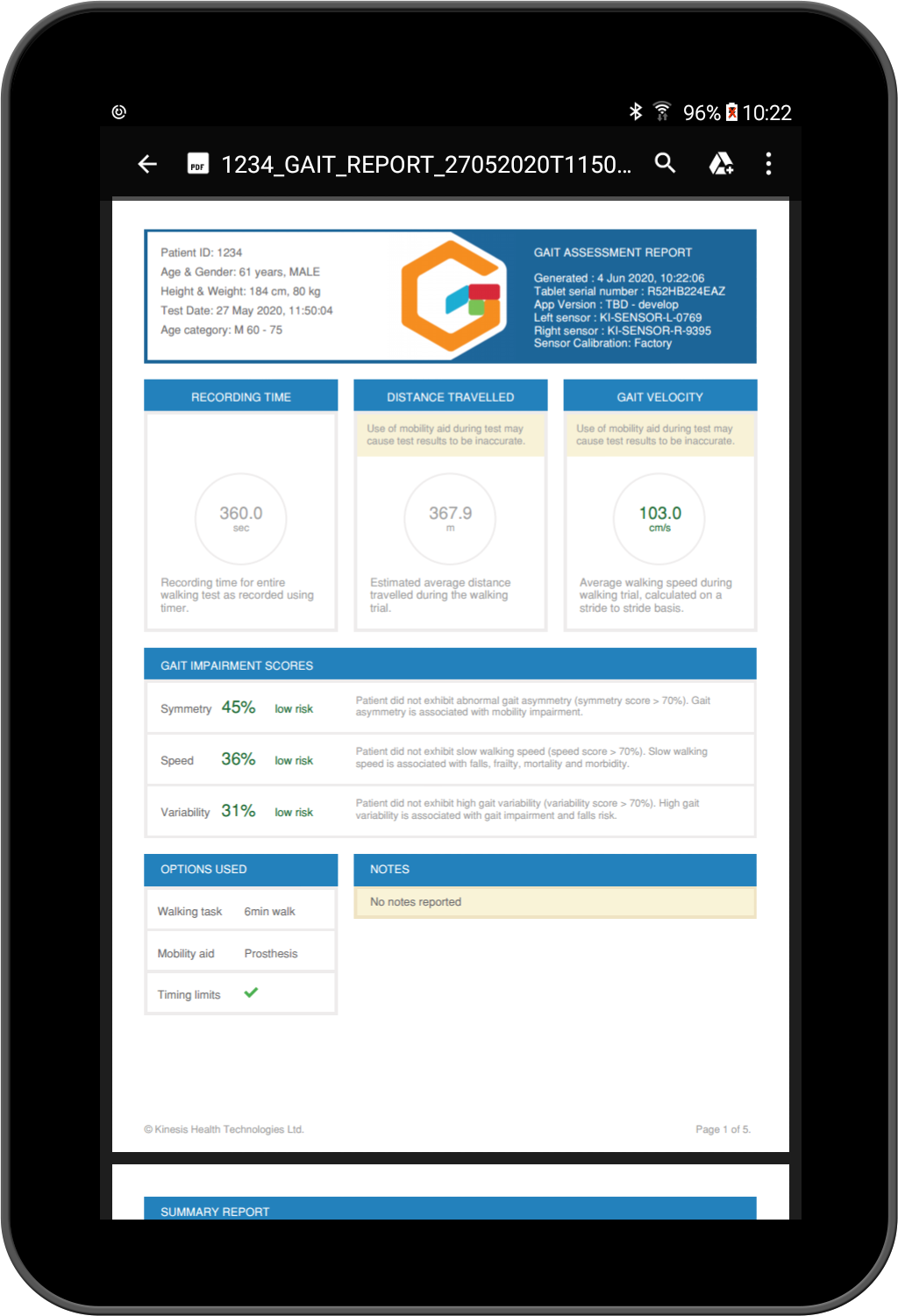
Create configurable reports on each gait test, in PDF and Excel formats. Export raw sensor data as well as individual stride data, add notes and tags on gait assessment conditions.
HIPAA and GDPR compliant - Securely and automatically back-up gait analysis results to the cloud.
Over 13 years of dedicated
scientific research have gone
into developing our Gait assessment platform

Fast, low-cost and portable assessment of gait

Using wearable inertial
sensors for accurate
gait analysis and assessment of mobility
GDPR/HIPAA compliant technology

Integrated, secure solution to
backup gait analysis data to remote
server via Wi-Fi
For ease of reference
and onward referral
Product is GDPR compliant and is a registered Class I medical device in the EU
(under MDR 2017/745) and carries the CE mark.
Kinesis Gait™ is HIPAA compliant and registered with the FDA as a Class I (exempt) medical device in the USA,
is registered as a Class I medical device with Health Canada,
and is registered with the Therapeutic Goods Administration (TGA) in Australia.
Product has been certified for electrical safety to medical device safety standard EN 60601-1.